John dalton atomic theory date4/3/2024 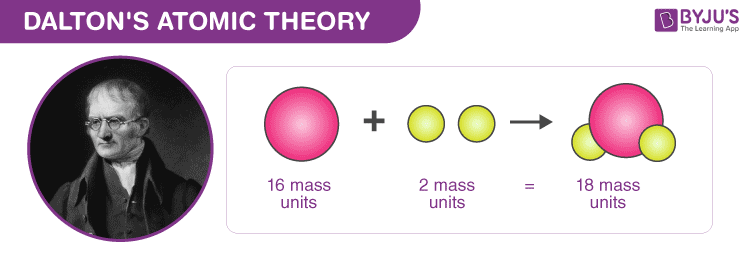
Fourth postulate: This postulate describes the combination of atoms which further form compounds and atoms of different elements which combine with each other to always combine in whole ratio number.ĥ. 
Complex compounds are the combination of atoms which will combine to form matter and atom is said to be the minimum unit for matter.Ĥ. Atoms are similar to conservation of mass like mass atoms are not created or destroyed. it cannot be divided into further smaller particles as it is the smallest unit even in the presence of chemical reactions. Third postulate: Third postulate of Dalton’s atomic theory states that atoms are of indivisible nature i.e. Second postulate: Second postulate states that atoms of the same element are exactly similar to each other in weight as well as the other properties, different elements have different types of atoms.ģ. 
It is also known that elements are the combination of atoms and these types of particles will not show any change unless they undergo some chemical reaction.Ģ. First Postulate: As discussed in the Dalton atomic model, atoms are indivisible in nature and these are also indestructible and have homogeneous tiny sphere like shape. There are six main postulates of Dalton’s atomic theory which can be described as follows:ġ. Just Study 40% Syllabus and Score upto 100% Download EBook State the postulates of Dalton’s atomic theory NCERT notes Class 11 Chemistry Chapter 2 Structure of Atom.NCERT Exemplar Class 11 Chemistry Solutions Chapter 2 Structure of Atom.NCERT solutions for Class 11 Chemistry Chapter 2 Structure of Atom.The elements which contain different types of atoms have different weights from each other. Same elements contain the same kind of atoms while different elements contain different types of elements.ħ. This theory also suggested that matter is composed of very tiny particles known as atoms.Ħ. Atoms of two or more elements interact with each other and form molecules.ĥ. This model is considered as the basic unit of all the matters.Ĥ. This name derived from the nature of the model as this model is made up of atoms which are indivisible and solid in nature and have the shape of a compact sphere.Ģ. The Dalton model is also known by some other name called spherical model. The main postulates of Dalton’s atomic model are given as follows:ġ. Write the postulates of Dalton's atomic theory. This model was discovered in 1803 and this was said to be the first atomic model. This model is also known by the name modern atomic theory or atomic postulates. Thus known as the John Dalton atomic model. This model is given by the scientist John Dalton. The first structural, conceptual and arrangement of atoms were proposed by a model called Dalton’s atomic model.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
